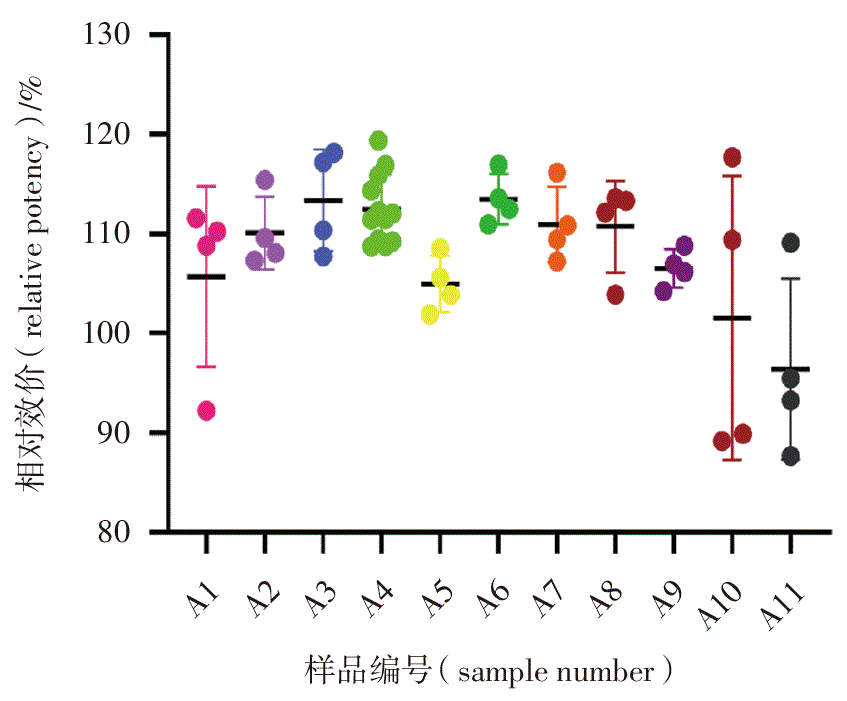
目的: 建立基于Nb2-11细胞增殖的聚乙二醇化重组人生长激素(PEG-rhGH)体外生物学活性测定标准化方法,研究Nb2-11细胞法替代体内动物法的可行性。方法: 利用Nb2-11细胞法检测9批PEG-rhGH注射液体外生物学活性,拟订实验有效标准。按照2020年版《中华人民共和国药典》通则9401进行方法学验证。4家实验室协作研究检测2批PEG-rhGH原液与9批注射液的生物学活性,研究方法的精密度。利用Nb2-11细胞法和体内动物法分别检测8批PEG-rhGH原液和9批PEG-rhGH注射液的生物学活性,研究体内外方法测定结果的一致性。检测24批不同效期PEG-rhGH注射液的体外生物学活性,研究PEG-rhGH注射液体外生物学活性的标准限度。结果: 本方法相对准确度的相对偏倚范围在-3.059%~3.557%,线性回归方程的斜率为0.984,中间精密度几何变异系数范围为5.667%~6.923%,拟订实验有效标准通过率为100%,实验室间几何变异系数为2.281%,体内外检测结果的平均比率为0.938,不同效期PEG-rhGH产品的相对效价在100%~119%。结论: 本方法具有良好的实验室内及实验室间重现性,与体内动物法检测结果具有良好的一致性。本方法可作为测定PEG-rhGH产品生物学活性的标准化方法替代体内动物法应用于PEG-rhGH的质量控制和放行检验。
张孝明
,
李鹤洋
,
李懿
,
胡绍旺
,
李春燕
,
胡馨月
,
孙悦
,
梁成罡
,
李晶
. PEG化重组人生长激素生物学活性测定方法的联合验证*[J]. 药物分析杂志, 2025
, 45(7)
: 1197
-1204
.
DOI: 10.16155/j.0254-1793.2024-1191
Objective: To establish a standardized method for the in vitro bioactivity analysis of polyethylene glycol-conjugated recombinant human growth hormone (PEG-rhGH) based on Nb2-11 cell proliferation and to study the feasibility of replacing in vivo animal bioassay with the Nb2-11 cell bioassay. Methods: The in vitro biological activity of 9 batches of PEG-rhGH injection was detected by the Nb2-11 cell method to draft the effective experimental standard. The method was validated in accordance with the General Chapter 9401 of the 2020 Edition of the Chinese Pharmacopoeia. Four laboratories collaborated to detect the biological activity of 2 batches of PEG-rhGH drug substance and 9 batches of injection to study the precision of the method. The biological activity of 8 batches of PEG-rhGH drug substance and 9 batches of PEG-rhGH injection was detected by the Nb2-11 ell method and in vivo animal method respectively to study the consistency of the results determined by in vitro and in vivo methods. The in vitro biological activity of 24 batches of PEG-rhGH injection with different expiration dates was detected to study the standard limit of the in vitro biological activity of PEG-rhGH injection. Results: The relative accuracy of the method showed a relative bias ranging from -3.059% to 3.557%. The linear regression slope was 0.984, with intermediate precision geometric coefficients of variation (GCV) of 5.667%-6.923%. The pass rate for the proposed effective experimental standards was 100%, the inter-laboratory geometric coefficient of variation was 2.281%, and the average ratio of in vivo and in vitro test results was 0.938. The relative potency of PEG-rhGH products with different expiration dates ranged from 100% to 119%. Conclusion: This method exhibits good reproducibility both within and between laboratories and showed good consistency with the in vivo animal bioassay. It can serve as a standardized alternative to animal testing for quality control and release testing of PEG-rhGH products.
[1] 张孝明,李晶,梁成罡.生长激素作用通路、调控机制及生物学活性测定研究进展[J].药物分析杂志,2022,42(11):1865
ZHANG XM,LI J,LIANG CG.Research progression on the action pathways,regulation mechanism and biological activity measurement of growth hormone[J].Chin J Pharm Anal,42(11):1865
[2] WANG Y,LANGLEY RJ,TAMSHEN K,et al.Long-acting human growth hormone receptor antagonists produced in E.coli and conjugated with polyethylene glycol[J].Bioconjug Chem,2020,31(6):1651
[3] MILLER BS,VELAZQUEZ E,YUEN KCJ.Long-acting growth hormone preparations-current status and future considerations[J].J Clin Endocrinol Metab,2020,105(6):e2121
[4] CHRISTIANSEN JS,BACKELJAUW PF,BIDLINGMAIER M,et al.Growth Hormone Research Society perspective on the development of long-acting growth hormone preparations[J].Eur J Endocrinol,2016,174(6):C1
[5] YANG Z,YAO Q,GONG L,et al.A superlong-acting growth hormone-polypeptide fusion for growth hormone deficiency treatment[J].Adv Healthc Mater,2023:e2302507
[6] YUEN KCJ,MILLER BS,BILLER BMK.The current state of long-acting growth hormone preparations for growth hormone therapy[J].Curr Opin Endocrinol Diabetes Obes,2018,25(4):267
[7] SHI Y,LU A,WANG X,et al.A review of existing strategies for designing long-acting parenteral formulations:focus on underlying mechanisms,and future perspectives[J].Acta Pharm Sin B,2021,11(8):2396
[8] SUN C,LU B,LIU Y,et al.Reduced effectiveness and comparable safety in biweekly vs.weekly PEGylated recombinant human growth hormone for children with growth hormone deficiency:a phase Ⅳ non-inferiority threshold targeted trial[J].Front Endocrinol(Lausanne),2021,12:779365
[9] LUO X,HOU L,LIANG L,et al.Long-acting PEGylated recombinant human growth hormone(Jintrolong)for children with growth hormone deficiency:phase Ⅱ and phase Ⅲ multicenter,randomized studies[J].Eur J Endocrinol,2017,177(2):195
[10] COX GN,ROSENDAHL MS,CHLIPALA EA,et al.A long-acting,mono-PEGylated human growth hormone analog is a potent stimulator of weight gain and bone growth in hypophysectomized rats[J].Endocrinology,2007,148(4):1590
[11] ESPOSITO P,BARBERO L,CACCIA P,et al.PEGylation of growth hormone-releasing hormone(GRF)analogues[J].Adv Drug Deliv Rev,2003,55(10):1279
[12] KARBASIAN M,KOUCHAKZADEH H,ANAMAGHI PN,et al.Design,development and evaluation of PEGylated rhGH with preserving its bioactivity at highest level after modification[J].Int J Pharm,2019,557(2):9
[13] 张孝明,黄盈,王绿音,等.利用Nb2-11细胞建立PEG化重组人生长激素生物学活性测定方法[J].药学学报,2023,58(3):773
ZHANG XM,HUANG Y,WANG LY,et al.A novel bioassay to assess bioactivity of PEG-rhGH using Nb2-11 cells[J].Acta Pharm Sin,2023,58(3):773
[14] ISHIKAWA M,NIMURA A,HORIKAWA R,et al.A novel specific bioassay for serum human growth hormone[J].J Clin Endocrinol Metab,2000,85(11):4274
[15] TANAKA T,SHIU RP,GOUT PW,et al.A new sensitive and specific bioassay for lactogenic hormones:measurement of prolactin and growth hormone in human serum[J].J Clin Endocrinol Metab,1980,51(5):1058
[16] 张孝明,黄盈,梁誉龄,等.基于Nb2-11细胞增殖的重组人生长激素生物学活性测定方法考察[J].中国医药工业杂志,2022,53(12):1726
ZHANG XM,HUANG Y,LIANG YL,et al.Investigation on biological activity assay of human recombinant growth hormone based on Nb2-11 cell proliferation[J].Chin J Pharm,2022,53(12):1726



