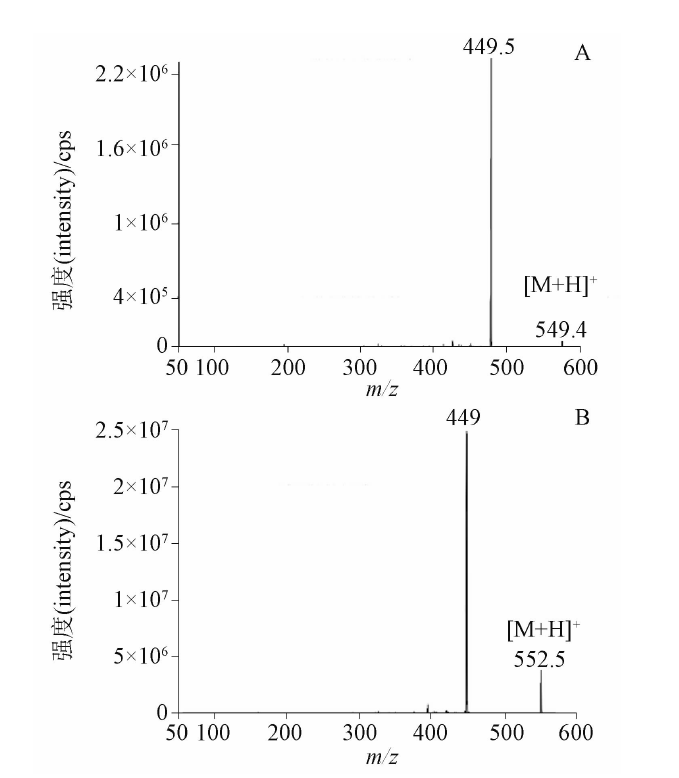
目的: 建立测定人血浆中FCN-437c药物浓度的HPLC-MS/MS方法,并用于FCN-437c的 Ⅰ 期临床研究。方法: 血浆经蛋白沉淀处理后,采用HPLC-MS/MS法进样测定。色谱柱为YMC Triart PFP柱(50 mm×2.1 mm, 5 μm),以0.5%甲酸水溶液(含5 mmol·L-1乙酸铵,A)-乙腈(B)为流动相,梯度洗脱,流速为0.5 mL·min-1,柱温为35 ℃,进样量为2 μL,进样器温度为10 ℃。质谱采用ESI+,MRM模式。检测离子反应对为m/z 549.4→449.5(FCN-437c)和m/z 552.5→449.0(FCN-437-D3,氘代内标),雾化气压力276 kPa,辅助气压力207 kPa,去簇电压100 V,碰撞室出口电压25 V。结果: 人血浆中FCN-437c的线性范围为5~1 000 ng·mL-1(r=0.999 0),定量限为5 ng·mL-1,批内、批间精密度分别小于2.0%和4.1%,平均回收率为104.0%(FCN-437c)、78.6%(FCN-437-D3),内标归一化基质因子为100%~102%。FCN-437c储备液4 ℃放置202 d,FCN-437c及FCN-437-D3工作液室温放置24 h,血浆样品室温放置20 h、冻融四循环、-20 ℃放置134 d、-80 ℃放置662 d,样品处理后自动进样器放置24 h,全血样品室温放置4 h均稳定。应用此方法检测了受试者口服FCN-437c后血浆药物浓度,ISR样品测试结果为97.0%与初测值的偏差在±20%以内。血浆样本稀释10倍后准确度为102.0%~108.0%。FCN-437c连续给药与单次给药相比,RAUC0-24和RCmax的累积比为1.33倍与1.59倍。结论: 本方法简便、准确、耐用、专属性强,可满足人血浆中FCN-437c的定量分析的要求。
Objective: To establish an HPLC-MS/MS method for the determination of FCN-437c in human plasma and its application to the phase I clinical study of FCN-437c. Methods: Following protein precipitation, plasma was injected and measured using HPLC-MS/MS method. The analytes were separated on a YMC Triart PFP column (50 mm×2.1 mm, 5 μm) using 0.5% formic acid (containing 5 mmol·L-1 of ammonium acetate, A) and acetonitrile (B) as the mobile phase with gradient elution at the flow rate of 0.5 mL·min-1, the column temperature was set at 35 ℃, the injection amount was 2 μL, and the injector temperature was 10 ℃. MS detection was performed with multiple reaction monitoring (MRM) mode using positive electrospray ionization. The ion transitions were m/z 549.4→449.4 for FCN-437c and m/z 552.3→449.3 for FCN-437-D3, respectively. Other mass spectrometry parameters were TEM, 500 ℃,GS1, 276 kPa,GS2, 207 kPa,DP, 100 V,CXP, 25 V. Results: The linear range of FCN-437c in human plasma was 5-1 000 ng·mL-1 (r=0.999 0). The lower limit of quantification was 5 ng·mL-1. The intra-batch and inter-batch precisions were less than 2.0% and 4.1%, respectively. The average recovery was 104.0% (FCN-437c), 78.6% (FCN-437-D3), and the internal standard normalized matrix factor was 100%-102%. The stock solution of FCN-437c was stable at 4 ℃ for 202 d, the working solution of FCN-437c and internal standard were stable at room temperature for 24 h. FCN-437c in human plasma was investigated to be stable at room temperature for 20 h, four cycles of freeze-thaw, -20 ℃ for 134 d, -80 ℃ for 662 d, as well as for 24 h in the autosampler after treatment. The whole blood samples were stable at room temperature for 4 h. This method was applied to the determination FCN-437c in human plasma, and the deviation between the test results and the initial values of 97.0% ISR samples was within ±20%. The accuracy was 102.0%-108.0% after 10-fold dilution of plasma samples. The cumulative ratio of RAUC0-24 and RCmax was 1.33 times and 1.59 times when FCN-437c was administered continuously compared with single administration. Conclusion: This method is simple, accurate, robust and specific, which can meet the requirements of quantitative analysis of FCN-437c in human plasma and also can be used to determine FCN-473c in human plasma of the phase I clinical study.
[1] BINOJ CN, RATNA KV. Regulation of hormonal therapy resistance by cell cycle machinery[J]. Gene Ther Mol Biol, 2008, 12:395
[2] DU Q, GUO X, WANG M, et al. The application and prospect of CDK4/6 inhibitors in malignant solid tumors[J]. Hematol Oncol, 2020, 13:41
[3] ADON T, SHANMUGARAJAN D, KUMAR HY. CDK4/6 inhibitors: a brief overview and prospective research directions [J]. RSC Adv, 2021, 47: 29227
[4] 常春, 王静萱. CDK4/6抑制剂治疗乳腺癌的研究进展[J]. 现代肿瘤医学,2021, 29(17): 3120
CHANG C, WANG JX. Research progress of CDK4/6 inhibitors in the treatment of breast cancer [J]. J Mod Oncol, 2021, 29(17): 3120
[5] GROENLAND SL, MARTÍNEZ-CHÁVEZ A, VAN DONGEN MGJ, et al. Clinical pharmacokinetics and pharmacodynamics of the cyclin-dependent kinase 4 and 6 inhibitors palbociclib, ribociclib, and abemaciclib [J]. Clin Pharmacok, 2020, 59: 1501
[6] FINN RS, MARTIN M, RUGO HS, et al. Palbociclib and letrozole in advanced breast cancer [J]. N Engl J Med, 2016, 375(20): 1925
[7] ORTOBAGYI GN, STEMMER SM, BURRIS HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer [J]. N Engl J Med, 2016, 375(18): 1738
[8] GOETZ MP, TOI M, CAMPONE M, et al. Abemaciclib as initial therapy for advanced breast cancer[J]. J Clin Oncol, 2017, 35(32): 3638
[9] STEFANO F, MARZIA DR, GIUSEPPE C, et al. Drug-drug interactions in breast cancer patients treated with CDK4/6 inhibitors [J]. Cancer Treat Rev, 2019, 74: 21
[10] ZHANG J, WANG XJ, WANG X, et al. Phase 1a study of the CDK4/6 inhibitor, FCN437c, in Chinese patients with HR+/HER2 advanced breast cancer [J]. Invest New Drugs, 2021, 39:1549
[11] AMITA P, ERIKA H, YAN X, et al. A phase Ⅰ dose-escalation and dose-expansion study of FCN-437c, a novel CDK4/6 inhibitor, in patients with advanced solid tumors [J]. Cancers, 2022, 14, 4996
[12] POETTO AS, POSOCCO B, GAGNO S, et al. A new dried blood spot LC-MS/MS method for therapeutic drug monitoring of palbociclib, ribociclib, and letrozole in patients with cancer [J]. J Chromatogr B, 2021,1185: 122985
[13] PAUL D, CHANDRAKALA P, SURENDRAN S, et al. Pharmacokinetic interaction study of novel combination of palbociclib and sorafenib for hepatocellular carcinoma in SD rats [J]. J Chromatogr B Analyt Technol Biochem Life Sci, 2019, 1108:25
[14] ASHISH K, PATEL YT, DAVIS A, et al. Development and validation of LC-MS/MS methods for the measurement of ribociclib, a CDK4/6 inhibitor, in mouse plasma and Ringer’s solution and its application to a cerebral microdialysis study [J]. J Chromatogr B, 2017, 1057: 110
[15] TURKOVIĆ L, BOKOR L, EKPENYONG O, et al. Development and validation of a novel LC-MS/MS method for the simultaneous determination of abemaciclib palbociclib ribociclib anastrozole letrozole and fulvestrant in plasma samples: a prerequisite for personalized breast cancer treatment [J]. Pharmaceuticals, 2022, 15:614
[16] WICKREMSINHE ER, LEE LB. Quantification of abemaciclib and metabolites: evolution of bioanalytical methods supporting a novel oncolytic agent[J]. Bioanalysis, 2021, 13(9):711
[17] Novartis Pharmaceuticals Corporation. 209092Orig1s000-Food and Drug Administration: NDA/BLA Multi-disciplinary Review and Evaluation NDA 209092 KISQALI (ribociclib) [DB/OL]. Center for Drug Evaluation and Research (2016-08-29) [2017-03-15]. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2017/209092Orig1s000MultidisciplineR.pdf
[18] 中华人民共和国药典2020年版.四部[S]. 2020: 466
ChP 2020.Vol Ⅳ[S]. 2020: 466



