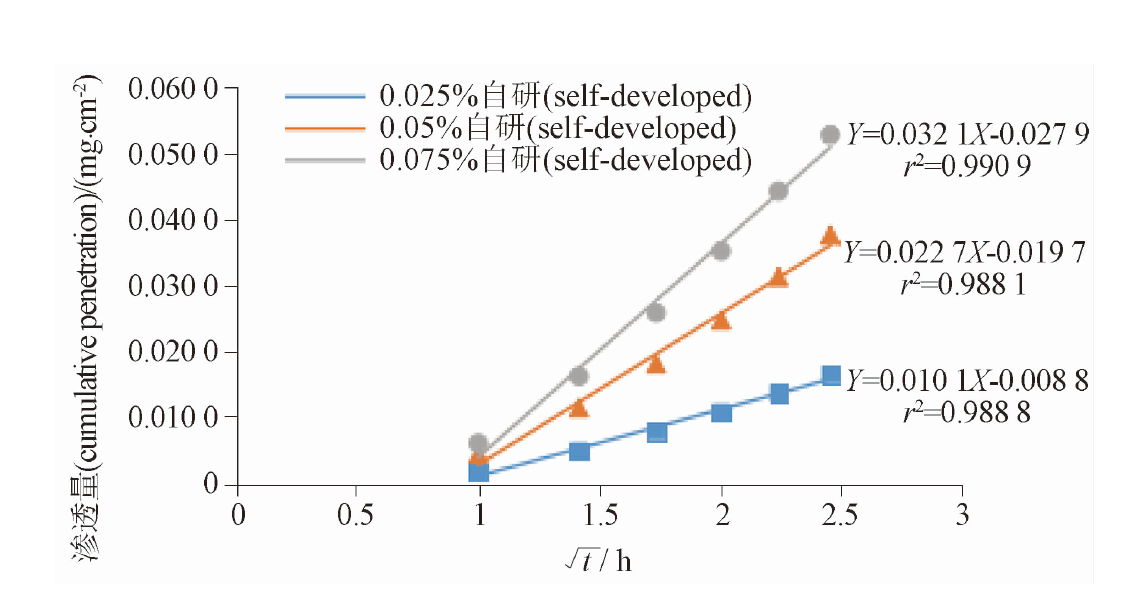
目的: 建立环孢素眼用乳剂体外释放方法。方法: 采用Franz扩散池,聚偏氟乙烯膜滤膜,以缓冲盐-无水乙醇(40∶60)为接收液,时间点为60、125、190、255、320、385 min。结果: 方法学验证表明,体外释放方法的滤膜惰性、专属性、灵敏度、选择性均符合规定。测定方法的定量限为0.07 μg·mL-1,在0.07~44.62 μg·mL-1范围呈现良好的线性关系,回收率为98.9%。依据FDA的判定原则,自研制剂与参比制剂体外释放行为一致。结论: 本法适合环孢素眼用乳剂的体外释放度评价。
Objective: To develop a method for characterizing in vitro release of cyclosporine ophthalmic emulsion. Methods: The Franz diffusion cell and the polyvinylidene fluoride membrane were adopted with buffer-ethanol (40∶60) as receiving media. The sampling time was set at 60,125,190,255,320,385 min, respectively. Results: The in vitro release method showed that the inertia of membrane, specificity, sensitivity and selectivity met the requirements. The validation of HPLC showed that the quantitative limit of the method was 0.07 μg·mL-1 and a good linear relationship between the concentration range of 0.07-44.62 μg·mL-1. The average recovery was 98.9%. Compared with the original preparation by FDA guideline, the in vitro release of the self-developed preparation was the same as the reference preparation. Conclusions: This method is suitable for the in vitro release evaluation of cyclosporine eye drops.
[1] 林琳,曹俊昌,杨政,等. 0.05%环孢素A纳米粒滴眼液治疗干眼症的实验研究[J]. 中医眼耳鼻喉杂志,2021,11(1):1
LIN L, CAO YC, YANG Z, et al. The treatment of 0.05%cyclosporine a nanoparticle eys drops on dry eye diease[J]. J Chin Ophthalmol Otorhinolaryngol, 2021,11(1):1
[2] 程林,张娴,李玉凤,等. 环孢素软胶囊在中国健康受试者中的生物等效性研究[J]. 中国临床药理学杂志,2022,38(4):342
CHENG L, ZHANG X, LI YF, et al. Bioequivalence of cyclosporine soft capsule in healthy Chinese subjects[J]. Chin J Clin Pharmacol, 2022,38(4):342
[3] FDA. Draft Guidance on cyclosporine[S]. 2016
[4] FDA. Draft Guidance on Acyclovir[S]. 2019
[5] FDA. Draft Guidance on Clindamycin Phosphate[S]. 2020
[6] FDA. Draft Guidance on Metronidazole[S]. 2019
[7] FDA. Draft Guidance on Tretinoin[S]. 2020
[8] FDA. Draft Guidance on Bexarotene[S]. 2019
[9] FDA. Draft Guidance on Docosanol[S]. 2017
[10] FDA. SUPAC-SS: Nonsterile Semisolid Dosage Forms; Scale-Up and Postapproval Changes: Chemistry, Manufacturing, and Controls; In Vitro Release Testing and In Vivo Bioequivalence Documentation[S]. 1997
[11] IFFNER K I, KANFER I, AMGMSTIN T, et al. A comprehensive approach to qμalify and validate the essential parameters of an in vitro release test (IVRT) method for acyclovir cream, 5[J]. Int J Pharm, 2018, 535(1/2): 217
[12] 胡昌勤,潘瑞雪. 溶出度试验评价/预测固体口服制剂生物等效性的研究进展[J]. 中国新药杂志,2014,23(1):44
HU CC, PAN RX. Progress in evaluation/prediction of bioequivalence of solid oral preparations by dissolution test[J]. Chin J New Drugs, 2014, 23(1):44
[13] 世界中医药学会联合会经皮给药专业委员会. 皮肤和黏膜用半固体制剂体外释放试验技术规范专家共识[J]. 中国现代应用药学,2022,39(20):2707
The Transdermal Drug Delivery Committee of the World Federation of Chinese Medicine Societies. Expert consensus on technical practice for the in vitro release test of topical semisolid dosage forms forskin and mucous membrane preparations[J]. Chin J Mod Appl Pharm,2022,39(20):2707
[14] 中华人民共和国药典2020年版. 四部[S]. 2020:132
Chp 2020. Vol Ⅳ[S]. 2020:132
[15] 石玲玲,陈轶嘉,朱云龙. 奥泽沙星皮肤外用制剂体外释放的对比研究 [J]. 药物分析杂志, 2023, 43 (11): 1989
SHI LL, CHEN YJ, ZHU YL. Study on in vitro pelease test conditions of the topical product of Ozenoxacin[J]. Chin J Pharm Anal, 2023, 43 (11): 1989
[16] 殷连珍,李慧敏,苏梅,等. 外用药物制剂体外释放试验技术要求概况 [J]. 中南药学, 2024, 22 (03): 741-747
YIIN LZ, LI HM, SU M, et al. Technical requirments of in vitro release test studies for topical drug products[J]. Cent South Pharm, 2024, 22 (3): 741
[17] 凌霞,叶晓霞,张弢,等. 苦参软膏体外释放评价方法的研究 [J]. 药物分析杂志, 2023, 43 (8): 1408
LING X, YE XX, ZHANG T, et al. Investgation on evaluation of in vitro release test for Kushen ointment[J]. Chin J Pharm Anal, 2023, 43 (08): 1408
[18] 邵鹏,郑金琪,潘芳芳,等. 外用半固体制剂的体外释放试验和等效性评价[J]. 中国现代应用药学,2021,38(20):2481
SHAO P,ZHENG JQ,PAN FF, et al. In Vitro release tests and equivalence evaluation for topical semisolid dosage forms[J]. Chin J Mod Appl Pharm, 2021, 38(20):2481