目的: 根据方法适用性试验结果,对微生态活菌制品的杂菌检查方法进行分类,并建立微生态活菌制品杂菌检查决策方法。方法: 按《中华人民共和国药典》(简称《中国药典》)四部<通则1105计数方法适用性试验>(简称<通则1105>要求),以大肠埃希菌、金黄色葡萄球菌、枯草芽孢杆菌、铜绿假单胞菌、白色念珠菌和黑曲霉为试验菌,采用胰酪大豆胨琼脂培养基、沙氏葡萄糖琼脂培养基、孟加拉红琼脂培养基、玫瑰红钠琼脂培养基,对已上市微生态活菌制品进行杂菌检查方法适用性试验。结果: 成分为厌氧菌或生长特性与试验菌差别较大微生物的制品,可参照《中国药典》四部<通则1105>进行方法适用性试验;成分中含有芽孢杆菌和肠球菌的制品,真菌计数可参照《中国药典》四部<通则1105>进行方法适用性试验;受成分菌的影响,非致病性杂菌无法通过方法适用性试验的产品,可采用选择性培养基对革兰阴性杆菌进行控制;无法通过计数方法适用性试验的制品,可根据给药途径、用药人群等,基于风险评估后,控制部分有潜在危害的微生物。结论: 本研究为微生态活菌制品的杂菌检查方法提供了选择依据,进一步补充完善了微生态活菌制品的质量标准体系。
Objective: To classify the microbiological examination methods of live microecological products according to the results of suitability test for the enumeration method, and establish the decision-making method for microbiological examination of live microecological products. Methods: The suitability test of the microbiological examination method of live microecological products was performed according to the requirements of the General Chapter 1105 in the Chinese Pharmacopoeia Vol Ⅳ. Escherichia coli, Staphylococcus aureus, Bacillus subtilis, Pseudomonas aeruginosa, Candida albicans and Aspergillus niger were used as the test strains. Tryptic Soy Agar, Sabouraud Dextrose Agar, Rose Bengal Agar and Rose Bengal Agar Ⅱ were used as medium. Results: For live microecological products composed of anaerobic bacteria or microorganisms with growth characteristics significantly different from the test strains, the suitability test for the method could be performed according to General Chapter 1105. For live microecological products containing Bacillus and enterococcus, the suitability test for the fungal enumeration method could be performed according to General Chapter 1105. Live microecological products could not be tested for suitability of the method due to the influence of the component microorganisms, gram-negative bacteria could be controlled using selective medium. In addition, some potentially harmful microorganisms could be controlled based on risk assessment according to the route of administration and the population of drug users. Conclusion: This study provides a basis for the selection of the microbiological examination methods for live microecological products, and further complements and improves the quality standard system of live microecological products.
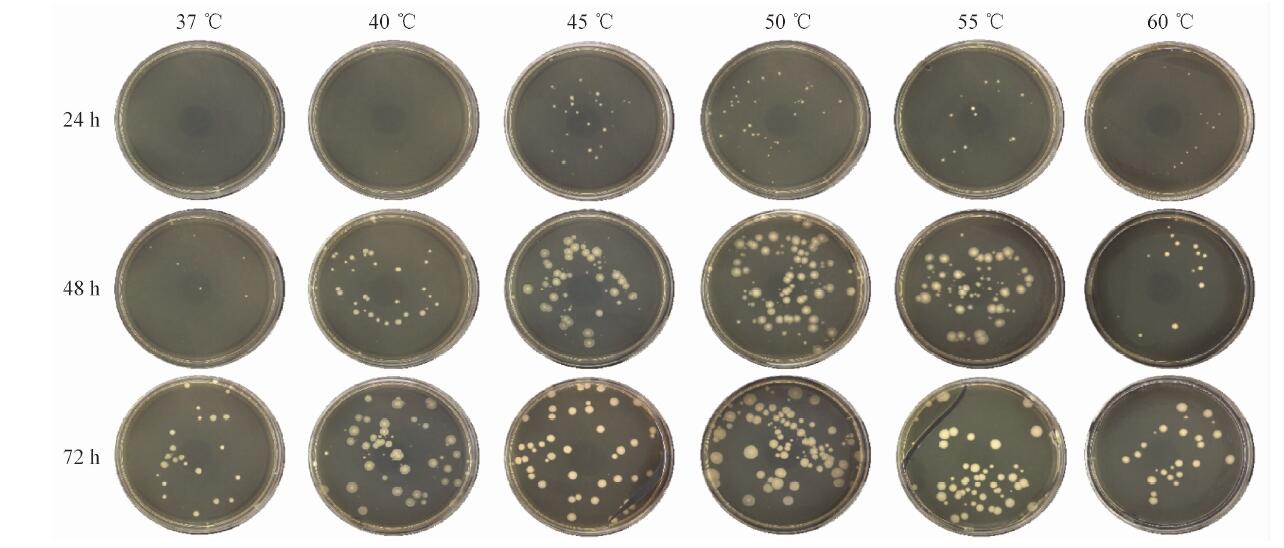
[1] 高倩,张宏翔,江洪,等.全球微生态药物研发现状及发展趋势[J]. 中国生物工程杂志,2020,40(Z1):166
GAO Q, ZHANG HX, JIANG H, et al. Current status and development trend of global microecological drug research and development[J]. Chin Biotechnol,2020,40(Z1):166
[2] ZHUO X, LUO HX, LEI R, et al. Association between intestinal microecological changes and atherothrombosis[J]. Microorganisms,2023,11(5):1223
[3] XING Y, GU X, RUAN G, et al. Probiotics for the treatment of gastric diseases[J]. Nutr Cancer, 2022,74(9):3051
[4] ZHANG H, WEN W, SHEN J, et al. Effect of microecological preparation supplementation on woman with polycystic ovary syndrome: a meta-analysis protocol[J]. Medicine,2018,97(44):e13040
[5] ZHU H, REN Z, ZANG Y, et al. Effects of microecological preparations on obese patients after bariatric surgery: a systematic review and meta-analysis[J]. Evid Based Complement Alternat Med, 2020(1): 8724546
[6] 赵紫楠,金鹏飞,李可欣,等.微生态制剂在呼吸系统疾病的研究进展及其在新冠肺炎中的应用价值[J]. 中国合理用药探索,2020,17(4):9
ZHAO ZN, JIN PF, LI KX, et al. Research progress of microecological agents in respiratory diseases and their application value in COVID-19[J]. Chin J Ration Drug Use,2020,17(4):9
[7] 张芷榕,周琴,薛炳乾,等.新型冠状病毒感染与肠道微生态的相关性研究[J]. 国际检验医学杂志,2021,42(20):2540
ZHANG ZR, ZHOU Q, XUE BQ, et al. Study on the correlation between 2019-ncov infection and intestinal microecology[J]. Int J Lab Med,2021,42(20):2540
[8] 韩芳,金小玲,刘晔,等.微生态制剂在2型糖尿病预防中的应用[J]. 医学综述,2021,27(1):153
HAN F, JIN XL, LIU Y, et al. Application of microecological agents in the prevention of type 2 diabetes mellitus[J]. Med Recapit,2021,27(1):153
[9] 许青. 通过全肠道菌群移植探究基于肠道菌群-肠-脑途径的阿尔茨海默病发病机制[D]. 成都: 西南交通大学, 2022
XU Q. Whole Gut Microbiota Transplantation to Explore The Pathogenesis of Alzheimer's Disease based on Gut Microbiota-Gut-Brain Pathway[D]. Chengdu: Southwest Jiaotong University, 2022
[10] 丁勃,沈振,任丽宏,等.基于基因功能分析的乳酶生与表飞鸣生产用菌株质量比对研究[J]. 中国新药杂志,2023,32(6):643
DING B, SHEN Z, REN LH, et al. Study on quality comparison of strains used in lactase production and Mefeiming production based on gene function analysis[J]. Chin J New Drugs, 2023,32(6):643
[11] 邢晟,王淑华,胡文红,等.益生菌制剂开发及相关质量标准构建策略[J]. 中国新药杂志,2021,30(9):773
XING S, WANG SH, HU WH, et al. Development of probiotics and related quality standard establishment strategy[J]. Chin J New Drugs, 2021,30(9):773
[12] 中华人民共和国药典2020年版. 三部[S]. 2020: 64 ChP 2020. Vol Ⅲ[S]. 2020: 64
[13] 鲁旭,田万红,马霄.微生态活菌制品质量控制[J]. 中国药品标准,2022,23(3):267
LU X, TIAN WH, MA X. Quality control of micro-ecological live bacteria products[J]. Drug Stand China, 2022, 23(3):267
[14] 中华人民共和国药典2020年版. 四部[S]. 2020: 161 ChP 2020. Vol Ⅳ[S]. 2020: 161
[15] 胡昌勤.药品微生物控制体系建设现状与展望[J]. 中国现代应用药学,2021,38(5):513
HU CQ. Current situation and prospect of microbial control system for drugs[J]. Chin J Mod Appl Pharm, 201,38(5):513
[16] USP-NF[S/OL]. 2023[2023-11-01]. https://doi.org/10.31003/USPNF_M11076_02_01
[17] EP 10.0[S]. 2019



